Latest News
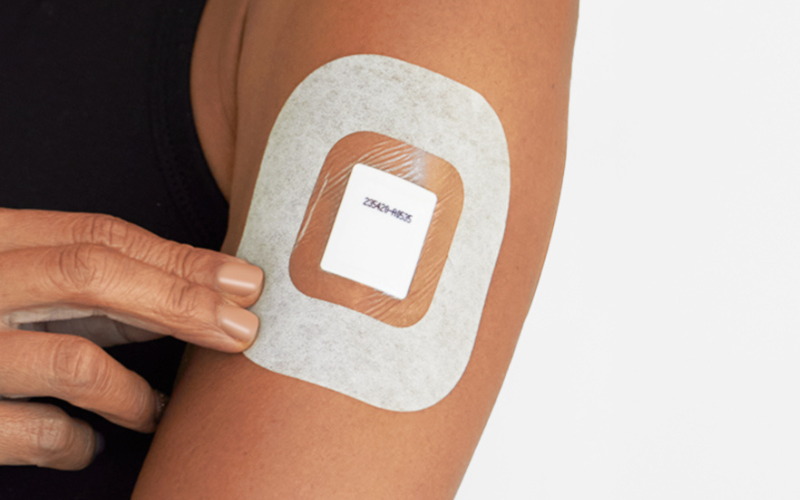
Intelligent Bio Solutions Adds Next-Gen Wearable for Extended Drug Detection to Product Offering, Strengthening Leadership in Non-Invasive Testing
NEW YORK, July 15, 2025 – Intelligent Bio Solutions Inc. (Nasdaq: INBS) (“INBS” or the “Company”), a medical technology company

Intelligent Bio Solutions to Present PK Study Data at Association for Diagnostics and Laboratory Medicine 2025 Clinical Lab Expo
Data shows excretion of codeine in fingerprint sweat INBS’ System is viable option for non-invasive diagnostic testing NEW YORK, July
Drug Test Detection Times: Fingerprint, Saliva, and Urine Compared
Understanding drug test detection times is essential when choosing a testing method that aligns with your organization’s safety and compliance
